Mass photometry of nanocages
Characterizing protein nanocages for nanomedicine applications
Protein nanocages, including ferritin, are integral in nature for various purposes like compartmentalization and cargo delivery. In healthcare applications, synthetic biologists often modify or design these nanocages for novel functionality.
However, characterizing stability and cargo loading of nanocages has traditionally posed challenges due to the limitations of available analytical techniques. Mass photometry emerges as a promising analytical technique to study protein self-assembly and nanocages. It directly measures the mass of biomolecules, providing label-free, rapid, and precise measurements with minimal sample consumption.
How to characterize ferritin assembly and reassembly?
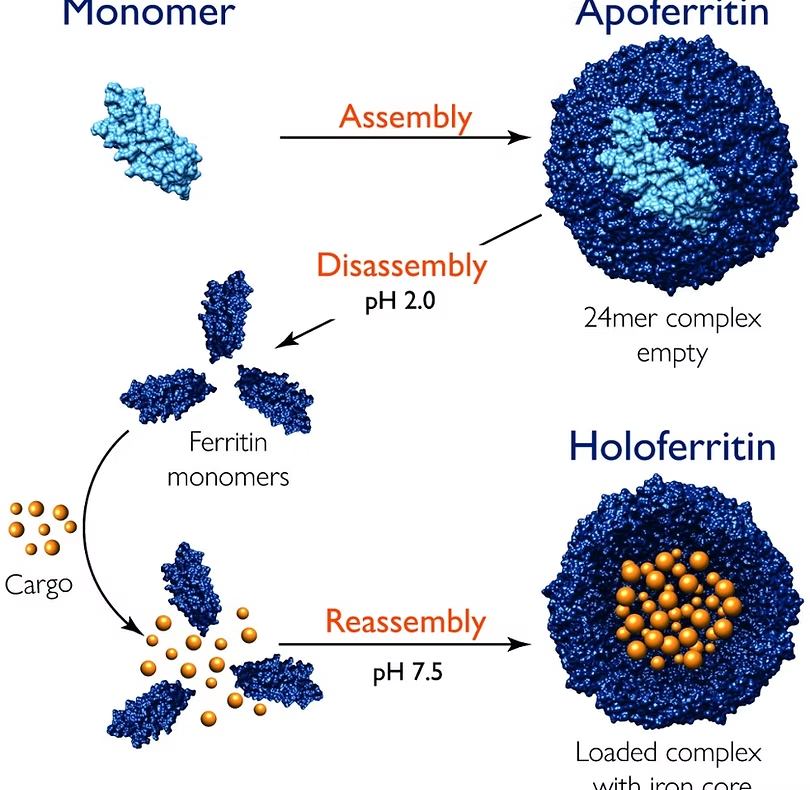
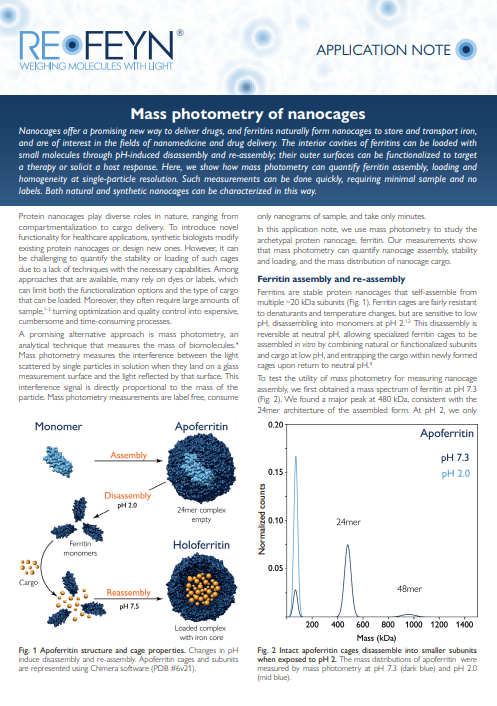
Focusing on the archetypal protein nanocage, ferritin, the application note referenced below demonstrates how mass photometry can effectively quantify nanocage assembly, stability, and pH-induced disassembly and reassembly. Ferritin, composed of multiple subunits, exhibits stability against denaturants and temperature changes but is sensitive to low pH, where it disassembles into monomers. Mass photometry enables precise monitoring of ferritin assembly at different pH levels.
Image: Changes in pH induce disassembly and reassembly of a nanocage. For experimental details, download the app note.
Mass photometry monitors nanocage load
Assessing the cargo loading of nanocages, such as iron-loaded ferritin, has traditionally been complex and time-consuming, often requiring techniques like atomic force microscopy or mass spectrometry.
Mass photometry was shown to reveal mass polydispersity in iron-loaded ferritin samples, a valuable insight for drug delivery applications. Mass photometry offers a quick, user-friendly, and label-free approach, eliminating the need for intricate sample manipulation, while consuming only minimal sample amounts. Mass photometry is poised as an efficient tool to rapidly assess the effect of buffer conditions on self-assembling proteins, stoichiometries, and incubation periods, and therefore supporting optimization of nanocage assembly and cargo loading protocols
To learn how mass photometry can help measure protein assembly in solution
Mass photometry of nanocages
Learn how mass photometry can be employed for the analysis of protein nanocages, such as ferritin. Discover how it can measure ferritin assembly, stability, and pH-induced disassembly/reassembly. Gain insights on how mass photometry revolutionizes the quantification of nanocage assembly dynamics, enabling quick and accurate assessments. Moreover, learn how it efficiently monitors nanocage cargo loading, revealing valuable insights for drug delivery applications.
Additional resources
APPLICATION NOTE: Formation of PROTAC ternary complexes measured with mass photometry
Learn how mass photometry can be used to inform on aspects of PROTAC ternary complex formation in solution, without requiring protein labelling or immobilization. These aspects involve a range of behaviors important for PROTAC mechanistic function: ternary complex formation, cooperativity, stoichiometry, and hook effect. Discover how you can apply mass photometry to visualize the possible intermediate interactions, due to cooperative effects, in a three-part PROTAC system.


WEBINAR: The evolution of carbon fixation: Studying Form I Rubisco using ancestral sequence reconstruction and mass photometry
Discover how mass photometry can be used in combination with ancestral sequence reconstruction to determine when an ancestral enzyme gained its structural complexity and how it started to interact with a novel subunit.
To discuss how mass photometry can be applied to the characterization of protein nanocages
More Application Notes
Browse through our catalogue of application notes highlighting some recent case studies featuring mass photometry.
