Rapid determination of stability by monitoring aggregation and degradation in a single measurement
Mass photometry is a revolutionary new method of analyzing molecules. It enables the accurate mass measurements of single molecules in solution, in their native state and without the need for labels. For molecular mass measurements with unmatched sensitivity, speed and simplicity of use, a TwoMP mass photometer offers wide mass range and single-molecule resolution.
Headline?
High-fidelity measurements of molecular mass
Minimal quantities of sample required
Intuitive acquisition and data analysis software
Easy setup – a compact, benchtop instrument with minimum installation requirements

With its high sensitivity, the TwoMP is ideally suited for measurements at physiological (i.e. low) concentrations. The high dynamic range intrinsic to single molecule counting techniques ensures low-abundance species are still captured accurately.
Refeyn’s software automatically controls the acquisition process and performs the mass analysis in minutes. You can intuitively interpret the mass distribution results without the need for any prior knowledge.
A compact, bench-top instrument with minimal installation requirements. Measurements only require a few microliters of sample and clean sample-carrier slides.
Automated mass photometry combines the ease and efficiency of automation with the unmatched sensitivity and simplicity of Refeyn’s molecular mass measurement technology. The automated mass photometer frees up operator time and offers enhanced precision, enabling rapid measurement of multiple samples with low sample consumption. Automation is compatible with the TwoMP.
Headline?
Rapid, automated measurement of multiple samples
Ideal for screening and titration assays
Highly reproducible data with a user-friendly software
Compatible with TwoMP unit



A user-friendly software interface
Automation unit compatible with TwoMP
Ideal for screening and titration assays
Precision pipetting
Consistent measurements
A user-friendly software interface
Automation unit compatible with TwoMP
Start where you are today and scale as you grow – both configurations deliver the same trusted mass photometry results.

For researchers who:

For teams who:

For researchers who:
Antigen-antibody interactions are a prime example of molecular systems that can be studied using mass photometry, which can be applied to determine binding affinities for mono- and multivalent interactions.
Mass photometry can be used to characterise DNA-protein complexes, which are crucial in gene expression, replication and DNA repair.
In this example, mass photometry not only allows detection of DNA binding but also provides information on how the oligomeric state of the protein changes upon DNA binding (Figure 2).
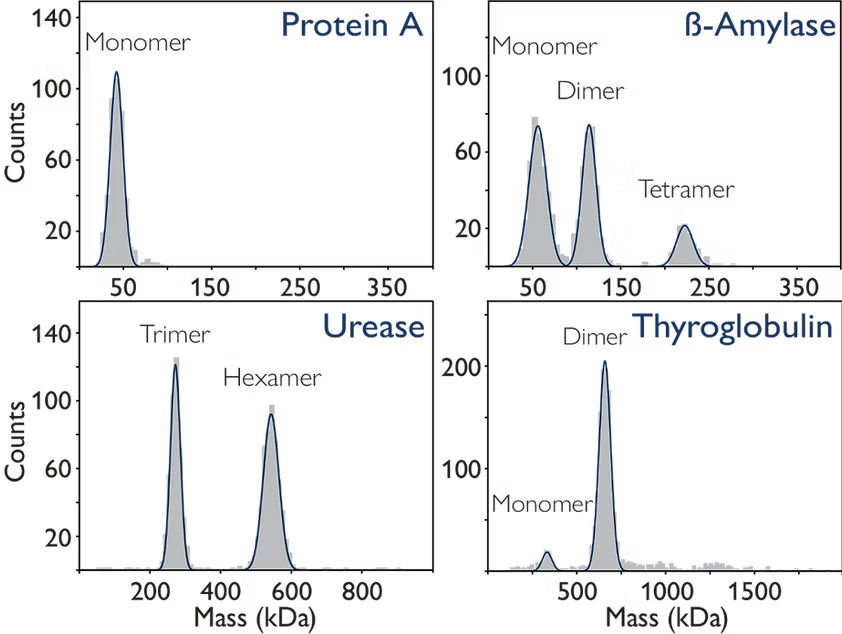
Many proteins adopt a particular oligomeric form under certain conditions. In Figure 1, mass photometry characterises four different proteins: protein A, beta-amylase, urease and thyroglobulin. Through this characterisation, these proteins showcase a range of behaviours – from purely monomeric (as in Protein A) to a dynamic equilibrium between multiple states.
Ribosomes are macromolecular assemblies of protein and RNA, and are central sites for protein synthesis. Bacterial ribosomes (in this example from E. coli) are >2 MDa in size. Magnesium ions play a subtle yet important role in the assembly of intact (70S) complexes. In the absence of magnesium, E. coli ribosomes rapidly disassemble into their 30S and 50S subunits. Mass photometry allows us to monitor these processes.
The liquid-handling robot autonomously analyses 24 samples in as little as 90 minutes in a highly reproducible manner.
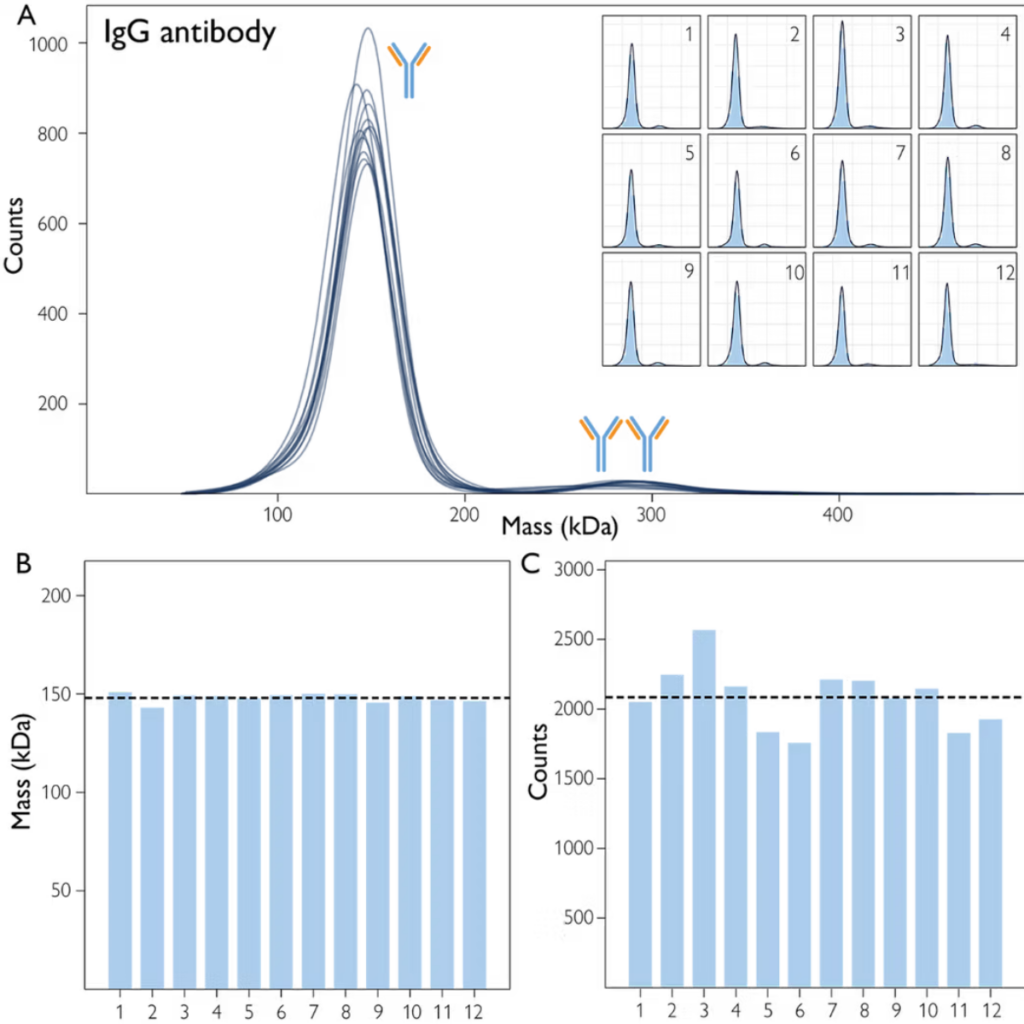
A straightforward, repeated measurement of a sample containing the antibody IgG illustrates the precision of the automated mass photometer, which is reflected in the high degree of reproducibility of the resulting data (Fig. 1).
Figure 1A shows histograms of 12 repeated measurements of an IgG sample made using automated mass photometry. Across the wells that contain the sample, the measured mass (Fig. 1B) and total counts (Fig. 1C) show low variability.
The TwoMP has a wide mass range, allowing the study of different biomolecules, including proteins, DNA, RNA, large macromolecular complexes, nanostructures and even small viruses such as AAVs.
The only optimization step necessary prior to a mass photometry measurement is to adjust the final concentration of the sample; the sample concentration on the instrument should be between 100 pM and 100 nM. The optimal concentration range for a mass photometry measurement is 5-20 nM.
For biomolecules within the mass detection range of the TwoMP (30 kDa to 5 MDa), the shape/conformation doesn’t influence the interferometric contrast and thus mass measured by the mass photometer.
In mass photometry, the mass measurement depends on the interference between the light scattered by the molecule and reflected light from the glass surface. This interference signal will be orders of magnitude greater than the fluorescence signal, so the mass measurement will not be affected.
If you already have a TwoMP, and SamuxMP, these mass photometers can be upgraded to automated systems. New automated mass photometers are available for purchase as TwoMP Auto and SamuxMP Auto.
We recommend using the Refeyn mass photometry consumables for Automated Systems. If you have a new TwoMP Auto instrument, you can order a Starter Kit, which includes everything you need to prepare samples and run a measurement (sample carrier slides and sample well cassettes to support 120 sample measurements, and the Alignment Assist Package to affix the sample well cassette). You can top up your supply of consumables with the Sample Preparation Package, which includes ready-to-use sample carrier slides and sample well cassettes to support 300 sample measurements or the Sample Carrier Slide-Only Package for Automated Systems (based on your needs).
Yes, there is an ‘in-plate’ dilution capability, and the user can select the following options in the software (version R1 2023): 1/2, 1/4, 1/8, 1/10, 1/25, 1/50, 1/100, 1/250, 1/500, 1/1000.
In terms of mass photometry, these are the same as the mass photometer inside the ‘Auto’. For ‘Auto’ specific specs, please refer to the data sheet.
MassFerence P1 is a custom calibrant for the OneMP, TwoMP and TwoMP Auto, designed to calibrate mass photometry measurements of proteins with molecular masses ranging from 90 to 1000 kDa.
Refeyn’s consumables are recommended to ensure consistent measurements with minimum hassle. Click below to order!
MassFerence P1 enables accurate, reproducible protein mass calibration across 90—1000 kDa for OneMP, TwoMP, and TwoMP Auto.

MassFluidix HC Sample Prep Kit enables fast, automated dilution and precise mass photometry for reproducible analysis of macromolecular interactions.

MassFluidix HC Preparation Plate simplifies chip setup with fast, error-free connections while protecting the objective.

MassGlass NA Kit streamlines nucleic acid analysis with cationic-coated slides for direct, reproducible RNA and DNA mass photometry on TwoMP, eliminating extra prep and enabling faster, reliable results.

MassGlass UC Sample Prep Kits enable reliable AAV and protein analysis on SamuxMP and TwoMP with clean, ready-to-use slides for reproducible results.

MP Alignment Kit streamlines AAV and protein analysis with SamuxMP and TwoMP for precise, reproducible results.
Find out how mass photometry can transform your sample characterization. Whether you’re focused on drug development, discovery science, or manufacturing, Refeyn’s innovative technology provides the insights you need to accelerate your breakthroughs.
Mass photometry is a revolutionary new method of analyzing molecules. It enables the accurate mass measurements of single molecules in solution, in their native state and without the need for labels. For molecular mass measurements with unmatched sensitivity, speed and simplicity of use, a TwoMP mass photometer offers wide mass range and single-molecule resolution.

High-fidelity measurements of molecular mass
Minimal quantities of sample required
Intuitive acquisition and data analysis software
Easy setup – a compact, benchtop instrument with minimum installation requirements
With its high sensitivity, the TwoMP is ideally suited for measurements at physiological (i.e. low) concentrations. The high dynamic range intrinsic to single molecule counting techniques ensures low-abundance species are still captured accurately.
Refeyn’s software automatically controls the acquisition process and performs the mass analysis in minutes. You can intuitively interpret the mass distribution results without the need for any prior knowledge.
A compact, bench-top instrument with minimal installation requirements. Measurements only require a few microliters of sample and clean sample-carrier slides.
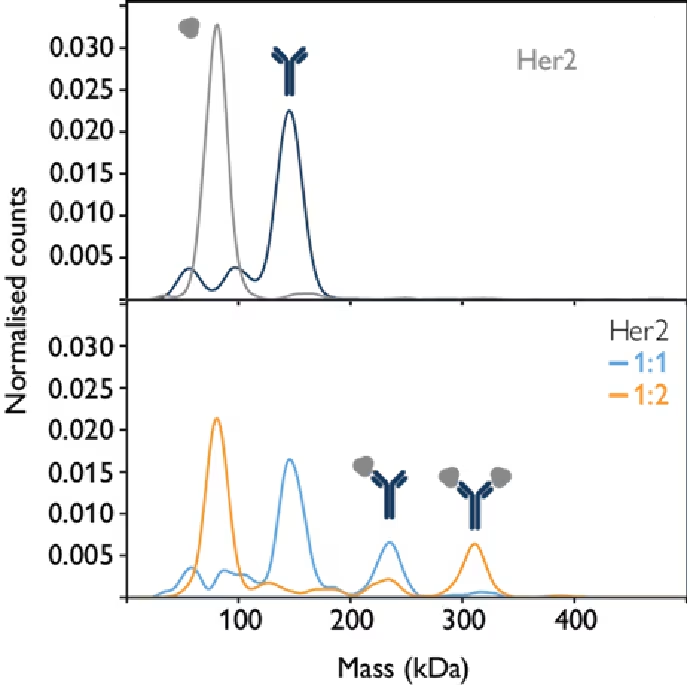
Antigen-antibody interactions are a prime example of molecular systems that can be studied using mass photometry, which can be applied to determine binding affinities for mono- and multivalent interactions.
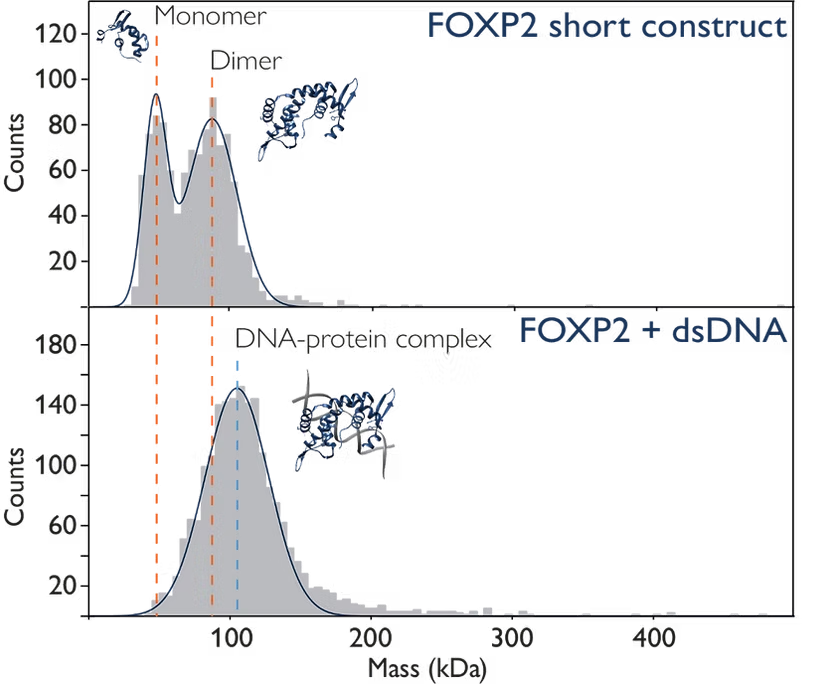
Mass photometry can be used to characterise DNA-protein complexes, which are crucial in gene expression, replication and DNA repair.
In this example, mass photometry not only allows detection of DNA binding but also provides information on how the oligomeric state of the protein changes upon DNA binding (Figure 2).
Many proteins adopt a particular oligomeric form under certain conditions. In Figure 1, mass photometry characterises four different proteins: protein A, beta-amylase, urease and thyroglobulin. Through this characterisation, these proteins showcase a range of behaviours – from purely monomeric (as in Protein A) to a dynamic equilibrium between multiple states.
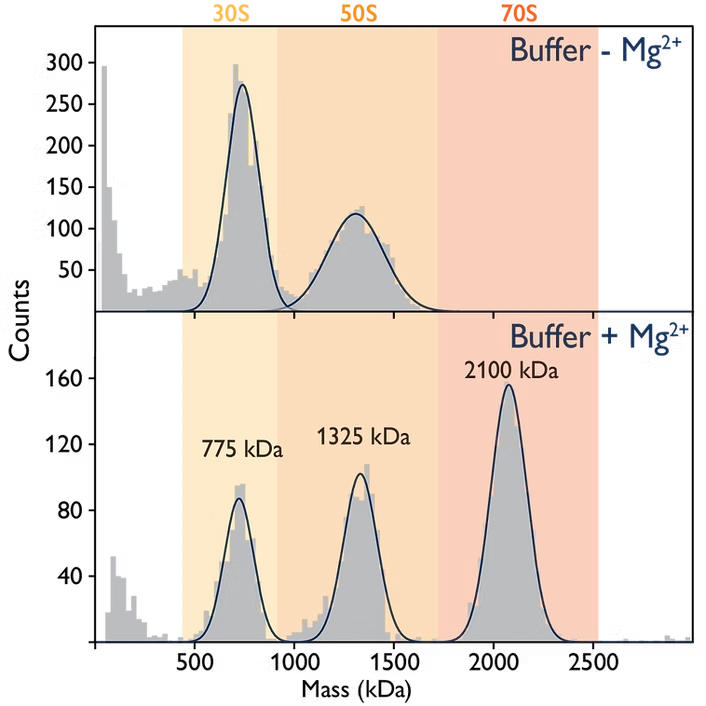
Ribosomes are macromolecular assemblies of protein and RNA, and are central sites for protein synthesis. Bacterial ribosomes (in this example from E. coli) are >2 MDa in size. Magnesium ions play a subtle yet important role in the assembly of intact (70S) complexes. In the absence of magnesium, E. coli ribosomes rapidly disassemble into their 30S and 50S subunits. Mass photometry allows us to monitor these processes (Figure).
The TwoMP has a wide mass range, allowing the study of different biomolecules, including proteins, DNA, RNA, large macromolecular complexes, nanostructures and even small viruses such as AAVs.
The only optimization step necessary prior to a mass photometry measurement is to adjust the final concentration of the sample; the sample concentration on the instrument should be between 100 pM and 100 nM. The optimal concentration range for a mass photometry measurement is 5-20 nM.
For biomolecules within the mass detection range of the TwoMP (30 kDa to 5 MDa), the shape/conformation doesn’t influence the interferometric contrast and thus mass measured by the mass photometer.
In mass photometry, the mass measurement depends on the interference between the light scattered by the molecule and reflected light from the glass surface. This interference signal will be orders of magnitude greater than the fluorescence signal, so the mass measurement will not be affected.
Read this application note to learn how you can use the TwoMP mass photometer to characterize protein-protein interactions, quickly and easily. Discover how you can determine the relative abundance of each protein and the complexes they form in solution, using this label-free bioanalytical tool. Learn also how you can use the mass photometry measurements to calculate the equilibrium dissociation constant (KD) for each interaction.


Check out this webinar to learn how mass photometry compares with size-exclusion chromatography (SEC) in measuring antibody aggregation. In the showcased study, both techniques were used to measure the aggregation levels of a monoclonal antibody used to treat breast cancer and several of its biosimilars.
The pros and cons of mass photometry and SEC for biomolecular characterization are discussed, highlighting the factors that should be considered when analyzing and comparing data generated by these two techniques.
Rapid determination of stability by monitoring aggregation and degradation in a single measurement.

MassFerence P1 is a custom calibrant for the OneMP, TwoMP and TwoMP Auto, designed to calibrate mass photometry measurements of proteins with molecular masses ranging from 90 to 1000 kDa.
Refeyn’s consumables are recommended to ensure consistent measurements with minimum hassle. Click below to order!

For researchers who:

For teams who: