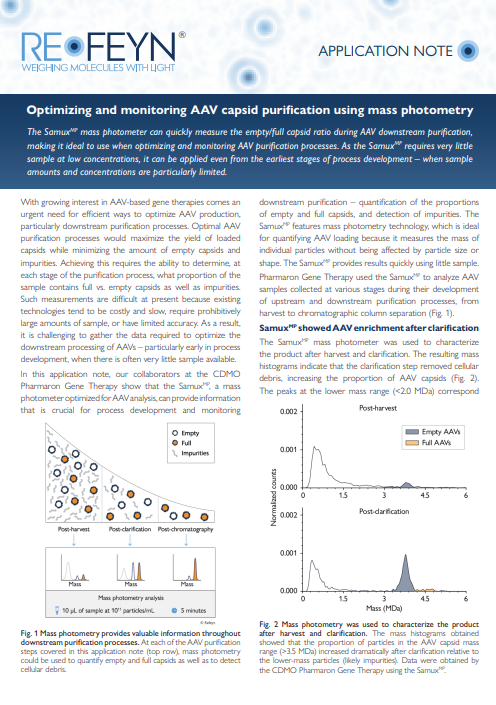
In this application note you will learn how mass photometry can be used to evaluate a sample at different stages during AAV purification, supporting AAV process development. You will see mass photometry analyses of samples collected at various stages during downstream AAV purification, including data on empty/full AAV ratios as well as on the presence of other impurities – such as cellular debris, partially filled and overfilled capsids, and aggregates. The data shown was obtained with the SamuxMP. Download the application note to learn the details.
Download the app note
Analytical techniques and challenges in AAV purification protocols
Adeno-associated viruses (AAVs) are a widely used vector for recombinant genome delivery into cells, and promising vehicles for novel gene therapies. The production AAV-based gene therapies require optimized purification processes that maximize the yield of loaded AAV capsids.

Mass photometry can support AAV purification protocols
Mass photometry is a powerful tool for AAV process development. Mass photometers like the SamuxMP and SamuxMP Auto are tailored for AAV analytics, providing comparable data to gold standard methodologies for AAV analytics with several advantages:
Accurate, single-particle characterization within the relevant mass range (0.5-6 MDa)
Detects impurities, heterogeneous capsid loading and aggregation in <5 minutes
Requires as little as 5-10 µL of sample with a concentration of 10^11 particles/mL
These strengths make mass photometry an ideal technique to support development and optimization of AAV purification protocols. This single-particle method detects even small quantities of impurities, and low turnaround times allows for multiple measurements at different stages of the AAV purification protocol. Finally, low sample requirements make mass photometry practical to implement in early process development, where AAV samples are small and valuable.
